  
N(t) = N_1\left(\frac $ which in this case is quite near to the mid hour. That is, the amount of coffee $N(t)$ for $t > 1$, is given by Then the amount of coffee left in your system, $t$ hours after you started drinking the coffee will be exactly what you already have in your question except the starting amount will be $N_1$ and you will subtract $1$ from the time in the function to account for the hour passed. Let $N_1$ be the amount of coffee in your system at $t = 1$ hours (when you are done drinking it). We split the time up into two cases: the first hour from when you begin drinking the coffee and then the rest of the time. CHEMISTRY The halflife of a radioactive substance is the time it takes for half of the atoms of. Q: Why is e special? (2.718., not 2, 3.Here is how we could approach this. Example 1 Exponential Decay of the Form y a(1 r)t.How To Think With Exponents And Logarithms.Understanding Exponents (Why does 0^0 = 1?).We then can say that N (t) N (1/2) (t/444) You asked what the constant value is for mercury 194. A Visual Guide to Simple, Compound and Continuous Interest Rates In this problem, we are given that it takes 444 years for the substance to lose 1/2 of its radioactive nuclei, so in each year, it will tick through only one-444th of its half-life.These are examples that model exponential growth and exponential decay. Demystifying the Natural Logarithm (ln) Formulas for half life and formulas for population growth. 
An Intuitive Guide To Exponential Functions & e.The natural log works on the ratio between the new and old value: $\frac$) implies you want people to think about change that happens at every moment.Įither way, be fluent in both models and learn to hop between the two. In our case, we grew from 1 to 2, which means our continuous growth rate was ln(2/1) =. The natural log finds the continuous rate behind a result. So, discrete changes can be modeled by some equivalent, smooth curve. 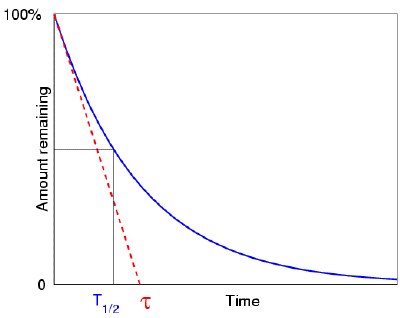
With enough splits, we could have smooth, continuous change. We can take them, split them into smaller, more frequent changes, and spread them out. I visualize change as events along a timeline:ĭiscrete changes happen as distinct green blobs. (Brush up on the number e and the natural logarithm.) Insight: Convert between discrete and continuous The pattern is always in motion (radioactive decay, a bacteria colony, or perfectly compounded interest). We can't point to an event and say "It changed here". 30-kg sample of Plutonium-239 will decay by 1 kg in 1,180 years. With continuous growth, change is always happening. Radioactivity and Half-Life Problems Using Calculus A. The mathematical representation of Half life is given by, (Half life time) (Napierian logarithm of 2)/(disintegration constant) The equation is: t 1/2 ln(2)/. Solution If 100 mg of carbon-14 has a half-life of. Example 1 Carbon-14 has a half-life of 5.730 years. The half-life of an isotope is the time taken by its nucleus to decay to half of its original number. Half-life is the time required for the amount of something to fall to half its initial value. The differential equation of Radioactive Decay Formula is defined as. A mating season finishes and offspring are born. It is the time requires to decay in half. We flip a coin and get new possibilities. Solution: Given decay constant 0.84 The half life formula can be used to find the half life of the substance. With n n being 17 years, the answer to your question is: 17 × log 5 log 2 39.4727776131 39 years 17 × log. Example 1: The decay constant of a substance is 0.84 s-1. Therefore, an answer to the following equation is sought: 0.2 1 2n 2n 1 0.2 5 n 2 log5 log 5 log 2 0.2 1 2 n 2 n 1 0.2 5 n 2 log 5 log 5 log 2. With discrete growth, we can see change happening after a specific event. A fraction of 20 grams of the initial 100 grams of isotope is 0.2. Note that the radioactive half-life is not the same as the average lifetime, the half-life being 0.693 times the average lifetime. The key question: When does growth happen? The predictions of decay can be stated in terms of the half-life, the decay constant, or the average lifetime.The relationship between these quantities is as follows.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
